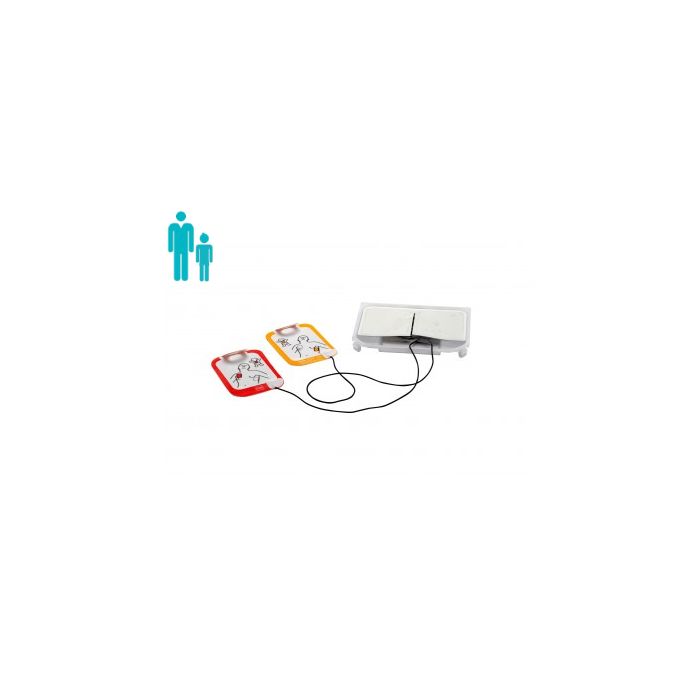
Physio Control Lifepak CR2 Replacement Electrode Kit (1 set)
This replacement Electrode Kit is for exclusive use with the Lifepak CR2 defibrillator. The CR2 is the latest in AED innovation from Physio Control and uses advanced INSIGHT™ technology to help increase chances of survival against cardiac arrest.
Key Features
- Up to 4 year shelf life
- Suitable for use with the Physio Control Lifepak CR2
As all electrode pads are single-use only, we always recommend you keep a spare pair in your rescue kit to ensure your device can always be used to save a life. With a 3-year shelf life, this replacement kit ensures your defibrillator is ready for use.
Thanks to the handy Paediatric switch on the CR2 device, these electrodes can be used both on Adult and Paediatric patients, this helps to reduce on-going costs as well as helping to provide faster treatment to the patient.
What's Included?
- Replacement Electrode Kit for Lifepak CR2 defibrillator
More information
For further information about this product, get in touch with a member of our team on 0161 776 7422.
| Defib Brand | Physio Control |
|---|---|
| Pad Life | Up to 4 years |
| FDA Approved | Yes |
| Dangerous Goods | No |