CU Medical Systems iPAD SP1 Semi Automatic Defibrillator
In stock
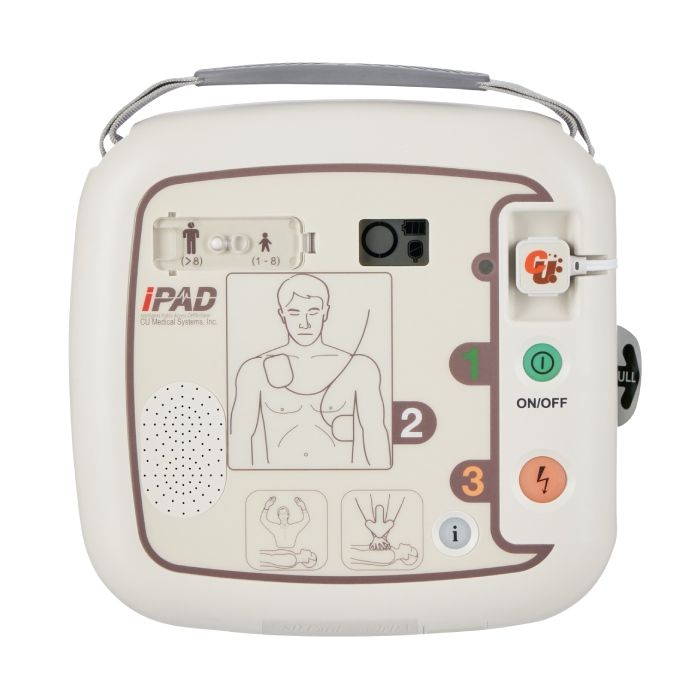
The iPAD SP1 is suitable for placement in a variety of environments. Adopting a simplistic design, this device enables both trained and untrained responders to act during a cardiac emergency, with confidence.
Key Defibrillator Features
- Paediatric Switch
- Integrated electrode-pad storage
- CPR detection technology recognises when CPR is being performed
- Visual indicators show battery life, unit status and pad status
- Internal memory stores up to 3 hours of data/last 5 cardiac events
- Any recorded data is easily transferable via SD Card and the IrDA ports
- Ambient-noise detector adjusts volume of audio instructions
- 7-Year warranty. When the device is registered, the warranty is extended by 3 years (link below)
- IP55 Rating
Suitable for placement in a variety of environments, the CU Medical System iPAD SP1 adopts a simplistic design allowing both trained and untrained responders to use the device and deliver treatment whilst feeling confident. The addition of the paediatric switch allows users to deliver treatment to children with ease, helping to save vital time and provide higher survival chances.
What's Included?
- 1 x CU Medical Systems iPAD SP1 Semi Automatic Defibrillator
- 1 x set of CU Medical Systems iPAD SP1 Adult Electrode Pads (Suitable for both Adults & Children)
- 1 x CU Medical Systems iPAD SP1 Disposable Li-ion Battery
- 1 x CU Medical iPAD SP1 Carry Case
- 1 x AED Rescue Kit
- 7 Years Manufacturer's Warranty - Extend to 10 years by registering here
Additional products available for this unit
More Information
For further information about this product, get in touch with a member of our team on 0161 776 7422.
| Connectivity | None |
|---|---|
| FDA Approved | No |
| Warranty | 7 Years Manufacturer's Warranty* |
| IP Rating | 55: the AED is protected against limited dust ingress and low pressure water jets from any direction |
| Defib Brand | iPAD (CU Medical) |
| Semi/Fully Automatic | Semi Automatic |
| Battery Life | 5 years |
| On Screen ECG | No |
| Energy Delivery | Fixed |
| Manual Override | No |
| Memory | 15 hours |
| Pad Life | Up to 3 years |
| Dimensions | H: 25.6cm x W: 26cm x D: 6.9cm |
| Product Weight | 2.4kg |
| Dangerous Goods | No |
| Icon | Label | Description | Type | Size | Download |
|---|---|---|---|---|---|

|
CU Medical Systems iPAD SP1 English Manual | User manual | 2.2 MB | Download |
The iPAD SP1 is suitable for placement in a variety of environments. Adopting a simplistic design, this device enables both trained and untrained responders to act during a cardiac emergency, with confidence.
More Information
For further information about this product, get in touch with a member of our team on 0161 776 7422.
Can users of all abilities use the CU Medical Systems iPAD SP1 Semi-Automatic Defibrillator?
The iPad SP1 defibrillator is designed to be a user-friendly device. During the rescue process, the iPad SP1 guides the user with calming voice prompts, clear instructions, and illuminated visual commands.
Each step of the way, users are given instructions, and the voice prompts automatically adjust to background noise. This prevents any inference with the treatment; otherwise, users may struggle to hear the device when noise increases.
What is an AED Rescue Kit?
All CU Medical Systems iPAD SP1 Semi-Automatic Defibrillators are complete with a CPR rescue kit. All the essential products are placed in a small green bag. The purpose of the rescue kit is to help prepare the casualty for treatment.
The rescue kit includes a breathing mask with oxygen connection, clothing scissors, gloves, a disposable razor, and alcohol wipes.
Do I need to purchase additional consumables with the CU Medical Systems iPAD SP1 Semi-Automatic Defibrillator?
Although the device has electrode pads suitable for adults and children, you can purchase SP1 child electrode pads separately. These are smaller pads to fit comfortably on a smaller frame but do not reduce the joule output. The child switch should still be used with paediatric pads on the iPAD SP1.
Does the iPAD SP1 defibrillator store data?
Yes, the iPAD SP1 defibrillator has internal storage that lasts up to 5 events and holds 3 hours of data. Using the built-in SD Card and IrDA ports, data is transferred and reviewed by medical professionals on a computer via the ‘CU-EX1’ software. The aim is to create a more streamlined casualty handover to advanced care.